Call for Service Provision on ”Advancing Personalised Oncology” – CLOSED
The canSERV Consortium announces its first Challenge-Call on “Advancing Personalised Oncology”. Cancer Researchers in, and outside the EU are invited to apply for free access to cutting-edge transnational services with an indicative overall budget of EUR 500,000. The aim of this call is to accelerate knowledge gain in personalised oncology with specific emphasis on multi-level data-based treatment guidance linked to Molecular Tumour Boards.
The projects supported under this call will support the advancement and implementation of personalised medicine strategies for all cancer patients in Europe, contributing to the overarching goals of the Cancer Mission Board.
Proposals for this call should address either of the two challenges listed below:
A.) Data-driven treatment selection for localised tumours with multiple patient-derived data types to support novel treatment and diagnostics.
Studying novel treatment and diagnostic options for localised cancer patients, e.g., colorectal cancer, typically involves highly diverse data sets (clinico-pathological variables, medical images, genomics and other omics, alternative experimental models, drugs, and clinical outcomes) collected either at one time point or repeatedly during therapy monitoring / follow-up. Services to obtaining such data should support either individual treatment guidance within a Molecular Tumour Board setting or facilitate a generalisable concept for innovative treatment selection of localized, non-metastatic tumours.
B.) Precision treatment guidance for metastatic cancer, especially colorectal cancer, based on multiple sources.
Different to the call above, this challenge addresses the metastatic disease stage with particular – but not exclusive – focus on colorectal cancer. The overall scenario and scope addressing novel treatment and diagnostic options using multi-modal data at single or multiple timepoints of individual patients or cohorts to foster knowledge gain for Molecular Tumour Board guidance applies like to the challenge call depicted under A). This could include interpretation of the functional relevance of gene alterations observed in a patient’s tumour and identification of standard-of-care drug biomarkers and investigational treatment opportunities
To address at least one of the two challenges with your research proposal, please apply for a combination of services provided by canSERV, ideally across the service domains (fields) in order to account for the intended multi-modal data approach. An extensive cross-domain service selection will be regarded as favourable for these two challenge calls.
The multi-dimensional data generated from these services for your specific research question will later on also be fed into federated data spaces to enable access, use, reuse and sharing of cancer-related data according to FAIR principles. This will allow to accelerate the possibilities for cross-European federated analyses of cancer-related data.
What is Transnational Access (TNA)?
Transnational Access allows researchers (users, applicants) free of charge access to services (equipment, expertise, resources) offered by the canSERV consortium members.
Transnational access can be:
- In person (physical access), with users physically visiting the research infrastructure facility/installation and receiving the service “hands-on”.
- Remotely (remote access), with research infrastructure resources and services offered without users physically visiting the facility/installation.
This opportunity is open to all European and non-European researchers.
Application
Application
Applications can be submitted by those individuals or organisations, who meet ALL the following criteria:
Who can apply?
Researchers (individual scientists, academic institutes, biotech/pharmaceutical SMEs, also group of scientists/networks/consortia) from public and private entities in European and non-European countries.
Young scientists and Small and Medium Sized enterprises are strongly encouraged to apply.
Affiliation
Applicants must be affiliated with an organisation in or outside the European Union. Up to EURO 100,000 of the indicative budget can be allocated to researchers outside the European Union.
Transnationality
Scientists can only apply for TNA services outside of their home country.
Dissemination
Results and/or data obtained from using the services provided under the canSERV Challenge Call must comply with the FAIR principles and will be shared initially with two ongoing initiatives within EU Cancer Mission, EOSC4Cancer and, when established, with UNCAN.eu, for the development and improvement of further services. Furthermore, the results and/or data will be reused within the canSERV project or other EU Cancer Mission initiatives at a certain time after service provision according to the canSERV user agreement. The data generated will then be included in the canSERV service list to support the canSERV Catalogue of Open Digital Research Services.
If the results and/or data are published, canSERV must be acknowledged in publications. Only user groups that are allowed to disseminate the results they have generated may be eligible for access (unless the users are working for SMEs).
Evaluation
Evaluation process
Applications will be evaluated by an international, independent review Panel and the Scientific Management Board of canSERV will approve the decisions.
The review Panel members will make their evaluation and recommendation by taking into consideration two criteria:
- Scientific merit and/or clinical impact of the research proposal
- Justification to the solicited service
Feasibility of the projects will be checked during the evaluation.
Applicants will be informed within 10 weeks after the submission deadline if their submitted proposal has been accepted.
How to apply
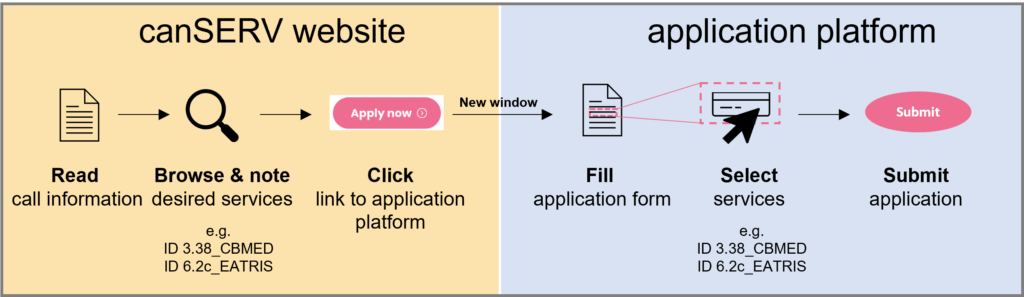
Two steps to get free-of-charge access to our services:
1 Browse our catalogue and identify the service/s you need.
2 Fill out the application form at the bottom of the page and hit the ‘SUBMIT’ button.
Please read the Terms & Conditions (for applicants)
For more information on our canSERV partner, please visit Partners.
Information about the Call, proposal submission and evaluation is available on request from calls@canSERV.eu
Catalogue of Services
Field 1 - Disease Models
These models range from state-of-the-art in vitro models like 3D cultures, organoids and organs-on-a-chip towards in vivo, patient-proximate models and advanced precision cancer models generated using CRISPR-Cas9.
Field 2 - Advanced technologies for Personalised Oncology
These services provide the cancer research community with access to cutting-edge services and technologies, as well as the associated expertise in their use, to address cancer research questions at a molecular, cellular and whole organism level.
Field 3 - Biomarker research, development, and validation
These services will provide access to cutting-edge and high-quality services (e.g., liquid biopsies, imaging biomarkers including radiomics, microbiomics, omics signatures, etc.) that will support the optimization of existing screening programmes, the advancement of novel approaches for screening and early detection, identification of new biomarker sets, as well as will contribute to the development of novel therapeutics based on molecular predictors.
Field 4 - New Therapeutic solutions
These services will provide access to a wide range of state-of-the-art services aiming to facilitate the development of novel therapeutic solutions for innovative personalised cancer research, as well as facilitate both clinical and translational research studies with focus mainly on: a) incorporate Small Molecules and Screening and b) Advanced Therapies and Biologicals Development Services.
Field 5 - Accelerated Translation into Personalised Oncology Clinical Practice
These services are specifically aimed at providing expert advice even to a single complex patient case or to a related finding or new discovery, so that patients can benefit from the results of cancer research possibly before the lengthy process of prospective clinical trials is completed.
Field 6 - Open Digital Research Services
These services aim to combine the complex landscape of reusable biomedical data and analytic tools in the context of tumour biology complexity, in order to provide unprecedented opportunities for the cancer community for the development of novel techniques and understanding of the impact of cancer diagnostics and treatment from a molecular to a population level.
Field 7 - Access to human samples and data
These services facilitate access to human biological samples and data. This will include provision of a) retrospective and on demand collections of high-quality biological material; b) high-quality associated data (i.e., OMICS, and clinical image data); and c) scientific advice on setting up state-of-the art related translational cancer research projects.
Training
Blindtext speaks the language of what it is designed for A Building must be sustainable to be beautiful (39)